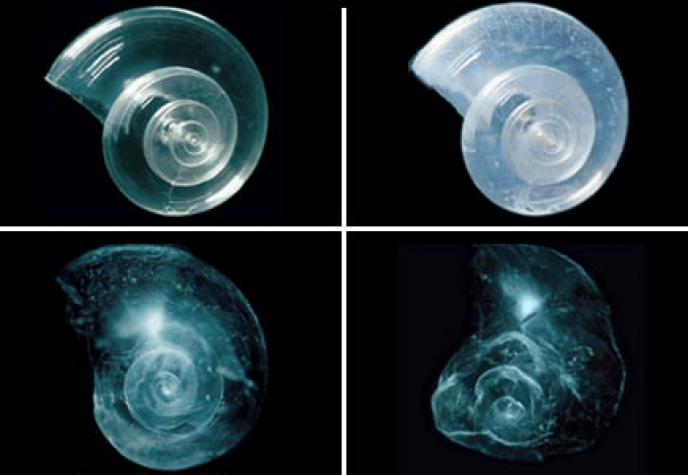
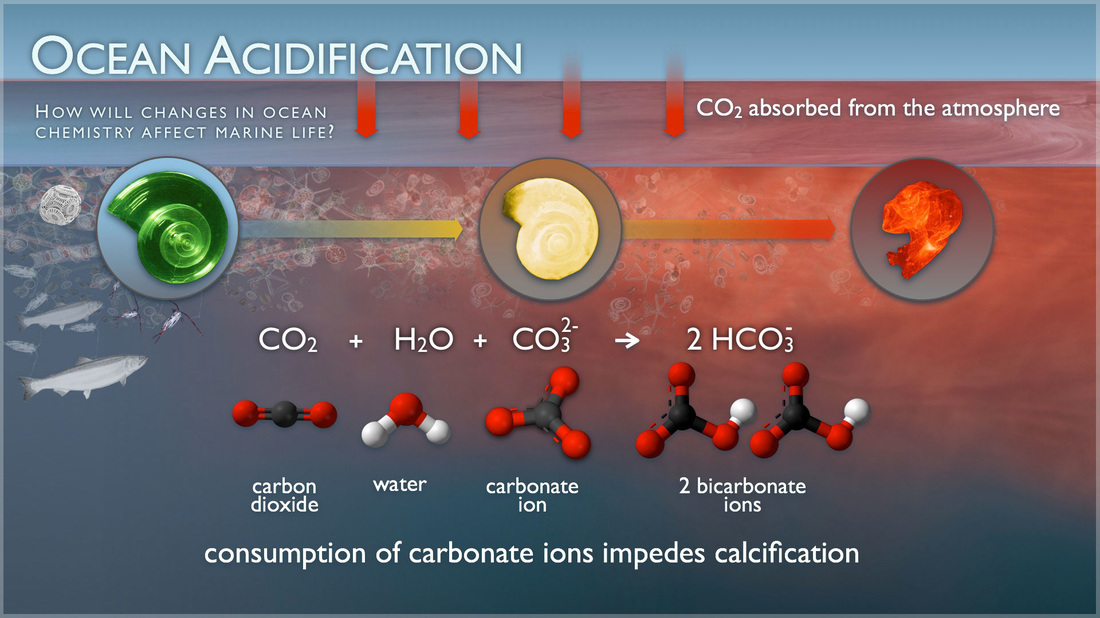
Ocean Acidification
Ocean acidification is damaging the marine ecosystem. Carbon Dioxide runoff from fossil fuels that humans burn is mixed in with the seawater and raises the acidity. This stunts the calcium carbonate growth on shells in the ocean, whether used by animals or not. It is our job to reduce this "acid pollution" and help out the creatures under the sea. It can be reduced or "re-balanced" back into equilibrium by using electric sources of fuel instead of fossil fuels. Also green energy (like wind farms and solar panels) are great ways to utilize and save the environment at the same time.
Chemical Equation
The chemical reaction CO2 + H2O <> H2CO3 works either way and describes the reaction with water and carbon dioxide to create carbonic acid. It is reversible as well. This is the process that breaks down the shells of ocean dwelling she'll creatures.
Statement
Ocean acidification an be stopped through the production of fuels that simply use more oxygen to create CO runoff instead of CO2. Most rocket engines use this type of fuel because they burn millions of gallons of fuels per liftoff. If cars could utilize this same type of technology the world would be a happier and more environmentally friendly place.
Reflection:
This project helped me understand a lot about chemical equilibrium. At first I did not understand exactly what this project was about but as it progressed I slowly understood more and more. I figured out that it needed to be a balance reversible reaction so my initial choice of rocket fuel was not doable. Then I changed to ocean acidification. I enjoyed this topic surprisingly more than I thought I would. Ocean acidification is reversible and it became very interesting to me. I would say that my group worked well but this was a singular project. I will say that m table group was extremely helpful with constructive criticism throughout this process. Overall I learned that I will should delve into things that I would not normally think about so I could find something new. I did really well at being efficient with time. Since at the last minute I changed topics I got my project together in a quarter of the required time. Overall I think this was a very well thought out project and I learned a lot about chemical equilibrium and ocean acidification.
This project helped me understand a lot about chemical equilibrium. At first I did not understand exactly what this project was about but as it progressed I slowly understood more and more. I figured out that it needed to be a balance reversible reaction so my initial choice of rocket fuel was not doable. Then I changed to ocean acidification. I enjoyed this topic surprisingly more than I thought I would. Ocean acidification is reversible and it became very interesting to me. I would say that my group worked well but this was a singular project. I will say that m table group was extremely helpful with constructive criticism throughout this process. Overall I learned that I will should delve into things that I would not normally think about so I could find something new. I did really well at being efficient with time. Since at the last minute I changed topics I got my project together in a quarter of the required time. Overall I think this was a very well thought out project and I learned a lot about chemical equilibrium and ocean acidification.